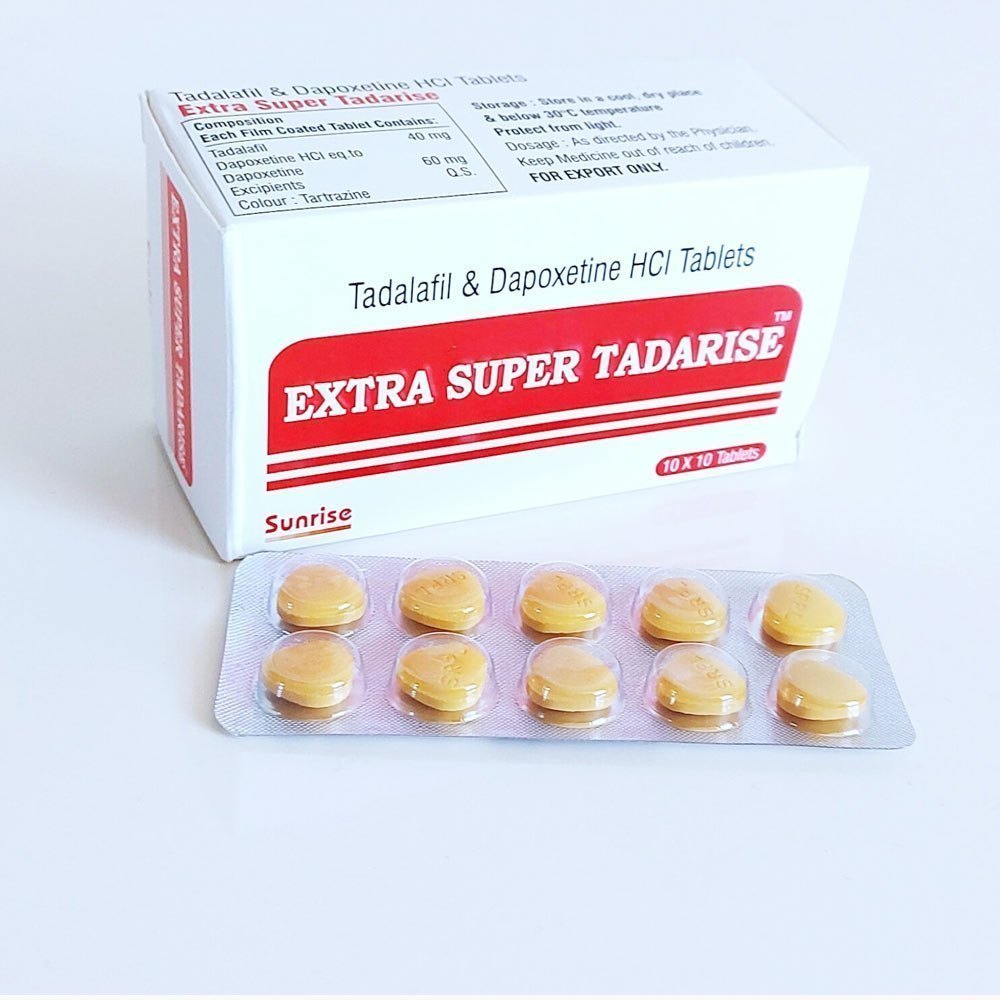
Description
Extra Super Tadarise Tablets are oral medications containing a combination of tadalafil (40 mg) and dapoxetine (60 mg). This dual-action formulation is designed to manage two common male sexual health conditions—erectile dysfunction and premature ejaculation.
Tadalafil is a phosphodiesterase type-5 (PDE-5) inhibitor that works by relaxing smooth muscles and increasing blood flow to the penile region during sexual stimulation, helping achieve and maintain a firm erection. Tadalafil is known for its long duration of action, with effects lasting up to 36 hours, allowing greater flexibility in sexual activity timing.
Dapoxetine is a short-acting selective serotonin reuptake inhibitor (SSRI) that increases serotonin levels in the nervous system, helping delay ejaculation and improve control over climax. This combination helps enhance sexual performance, stamina, and overall satisfaction.
Extra Super Tadarise Tablets are typically taken 30–60 minutes before sexual activity with water. Sexual stimulation is required for effectiveness, and the medicine should not be taken more than once within 24 hours.
Common Side Effects
-
Headache
-
Dizziness
-
Flushing
-
Nausea
-
Indigestion
-
Back pain
-
Nasal congestion
Important Warnings
-
Take only one tablet per day.
-
Avoid alcohol and recreational drugs.
-
Do not use with nitrate medications or other ED medicines.
-
Use cautiously in patients with heart, liver, kidney, or blood pressure disorders.
-
Intended for adult men only under medical supervision.


Reviews
There are no reviews yet.